Are you wondering if a biologic is indicated for chronic rhinosinusitis with nasal polyps? In this blog post, we’ll discuss the when and why of using biologics to treat this condition. We’ll also provide an overview of what to expect from the treatment, as well as other potential treatments. So, if you’re curious about using biologics for your chronic rhinosinusitis with nasal polyps, keep reading!
Introduction to Biologics for Chronic Rhinosinusitis with Nasal Polyps
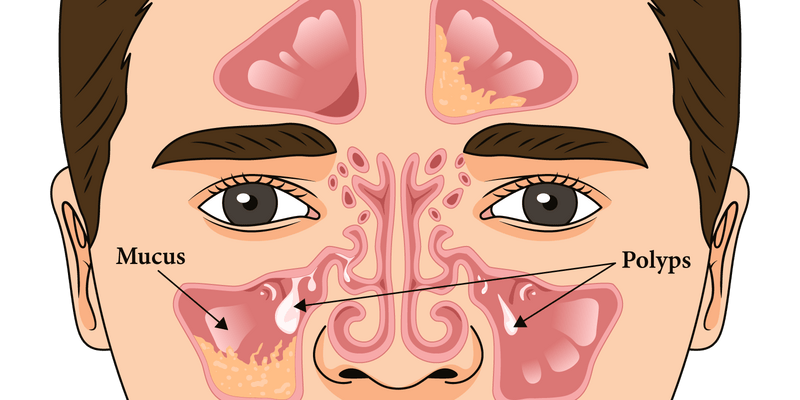
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a common condition that affects the nose and sinuses. It is typically treated with intranasal corticosteroids (INCS), saline irrigation, and systemic corticosteroids. Endoscopic sinus surgery is often needed to remove nasal polyps and open the sinus passages. However, in severe cases, biologics may be indicated. Biologics are a type of medication that target specific parts of the immune system to reduce inflammation, and they have been found to be effective in treating CRSwNP. Dupilumab, omalizumab, and mepolizumab are three biologics that are approved for treating CRSwNP. In this blog, we will discuss when biologics may be indicated for CRSwNP and how these medications can help patients.
Dupilumab, Omalizumab, and Mepolizumab: An Overview
Dupilumab, mepolizumab, and omalizumab are injectable biologic medications approved for patients 18 years and older with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP) as add-on therapy to intranasal corticosteroid sprays.
These medicines have been found to be beneficial in the treatment of CRSwNP, significantly reducing nasal polyp size, nasal congestion, and improving quality of life.
Dupilumab is a medication used to treat a number of inflammatory conditions, including moderate to severe atopic dermatitis (eczema), prurigo nodularis, asthma, eosinophilic esophagitis, and CRSwNP. It works by blocking proteins called interleukins (ILs) from binding to their cell receptors. Dupilumab particularly targets the two ILs, IL-4 and IL-13, which are thought to contribute to inflammation in atopic conditions. By blocking these interleukins from binding to their cell receptors, it also inhibits the expression of numerous other inflammatory markers that trigger symptoms. Dupilumab is administered every 2 weeks by subcutaneous injection as an add-on maintenance treatment to intranasal corticosteroids.
Omalizumab is an anti-IgE monoclonal antibody that binds to IgE in the blood, reducing the amount of free IgE available to bind to allergens. It is used to treat allergic asthma or CRSwNP and reduce the need for corticosteroids. Omalizumab is administered as an injection every 2-4 weeks.
Mepolizumab is an anti-IL-5 monoclonal antibody that blocks the action of IL-5, which is responsible for eosinophil maturation, survival, and activation. It is used to treat severe asthma and CRSwNP. Mepolizumab is administered as a injection once a month.
Biologics for CRSwNP
Biologic therapies are a new and effective treatment option for patients with chronic rhinosinusitis with nasal polyps (CRSwNP). Dupilumab was the first biologic to be approved by the FDA in June 2019 for adults with CRSwNP who were not adequately controlled by intranasal corticosteroids and saline irrigation. In addition, omalizumab and mepolizumab are also indicated for CRSwNP in patients 18 years of age and older.
Benefits of Biologics for CRSwNP
Biologics offer a number of benefits to those suffering from CRSwNP. They can reduce the need for oral steroids, which can cause serious side effects and can be used as an add-on therapy to intranasal corticosteroids and saline irrigation. Biologic drugs also come with fewer side effects, as they are highly targeted treatments, and many have been shown to significantly improve symptoms, polyps, and quality of life for patients with CRSwNP.
Risks and Side Effects of Biologics
The use of biologics for the treatment of chronic rhinosinusitis with nasal polyps is becoming more common, however, it is important to note the associated risks and potential side effects. Headaches, injection site reactions and pharyngitis are some of the most common adverse events reported in clinical trials. Additionally, biologics should be used cautiously as they may increase the risk of steroid side effects. Therefore, it is important to consult a healthcare professional if you experience any adverse events while taking biologics.
Conclusion
In conclusion, biologics are an effective and safe treatment for patients with CRSwNP who are 18 years of age or older. The three approved biologics, dupilumab, mepolizumab, and omalizumab, showed favorable results in clinical trials and can be used as add-on therapy to intranasal corticosteroids and saline irrigation for CRSwNP. It is important to discuss the benefits and risks of any biologic therapy with your healthcare provider in order to make an informed decision about treatment options.
SinusHealth promotes research, education, and advocacy for the care of sinusitis. Contact us with your questions and receive expert advice from board-certified Rhinologists.